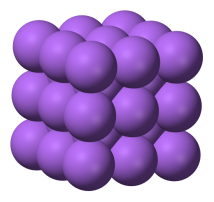
Solids, liquids and gases
solidsIn a solid the particles (ions, atoms or molecules) are closely packed together. The forces between particles are strong so that the particles cannot move freely but can only vibrate. As a result, a solid has a stable, definite shape, and a definite volume. Solids can only change their shape by force, as when broken or cut.
|
LiquidsA liquid is a nearly incompressible fluid that conforms to the shape of its container but retains a (nearly) constant volume independent of pressure. The volume is definite if thetemperature and pressure are constant. When a solid is heated above its melting point, it becomes liquid, given that the pressure is higher than the triple point of the substance. Intermolecular (or interatomic or interionic) forces are still important, but the molecules have enough energy to move relative to each other and the structure is mobile. This means that the shape of a liquid is not definite but is determined by its container. The volume is usually greater than that of the corresponding solid, the best known exception being water, H2O. The highest temperature at which a given liquid can exist is its critical temperature
|
gasesBlueberry Cobbler
Lorem ipsum dolor sit amet consectetur $7.00 Ice Cream Lorem ipsum dolor sit amet consectetur $4.00 |